
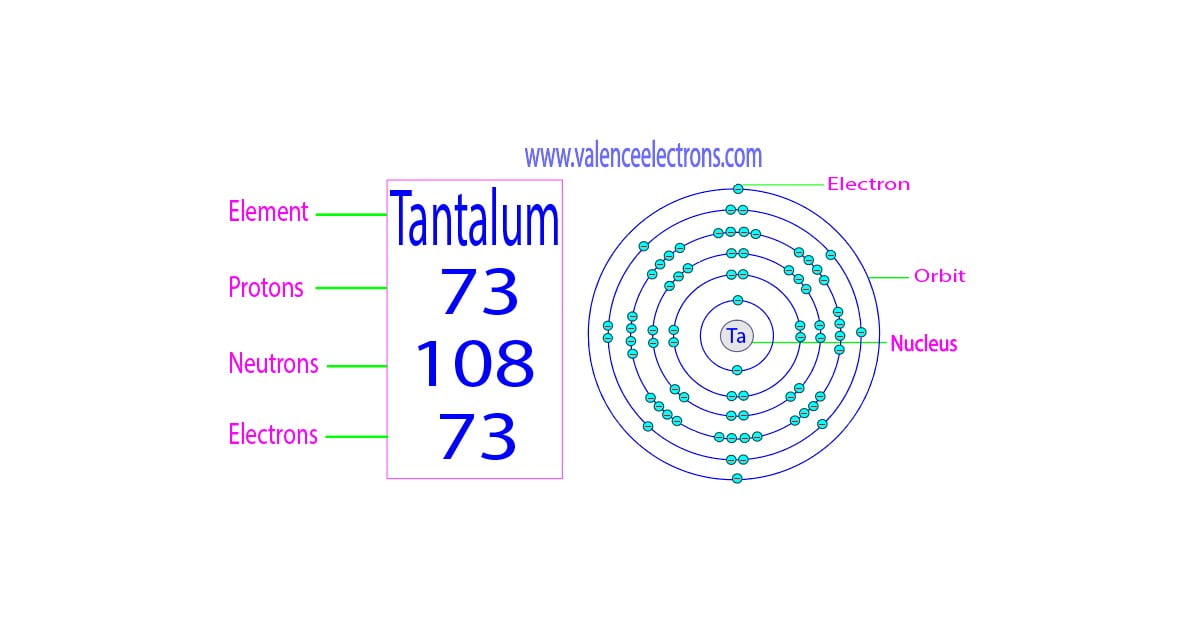
He found their maximum specific charge was around two thousand times smaller than that of the electron. Wien had used electric and magnetic fields to examine the positive rays. This shows that they could be massive, positively charged particles. They were much harder to deflect than cathode rays and were deflected in the opposite direction. The positive charge of the rays was not identified until sixteen years later when Wien deflected them in a magnetic field. These he called ' kanalstrahlen ’ or sometimes known as canal rays. In 1896 Goldstein noticed that when a discharge occurred in a tube of low-pressure gas rays appeared to be produced originating from the anode. Thus positive rays had first been observed by Goldstein in 1886. Ĭanal Ray experiment is the experiment performed by German scientist Eugen Goldstein in 1886 that led to the discovery of the proton. These rays were positively charged, and Thomson wanted to see if they were also composed of charged particles. In 1907 Thomson began to investigate the rays that were moving towards the cathode. They had previously been known as 'cathode rays' because in a vacuum tube they started at the negative electrical terminal called a cathode and moved away from it. Thomson referred to electrons as 'corpuscles'.
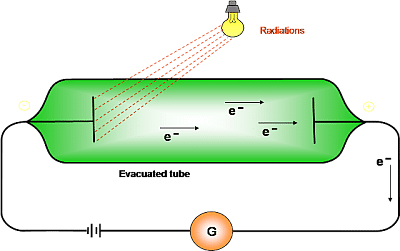
The electron is a tiny particle, almost two thousand times lighter than the hydrogen atom, and it is negatively charged. 1 POSITIVE RAYS-PRODUCTION AND PROPERTIES
